Imagine waking up tomorrow and discovering that the world around you has lost all its color. The vibrant red of a sunset, the lush green of summer leaves, the brilliant blue of the ocean all reduced to shades of black, white, and gray. For approximately one in thirty thousand people worldwide, this isn’t a hypothetical scenario. This is their reality from birth due to a rare genetic condition called achromatopsia.
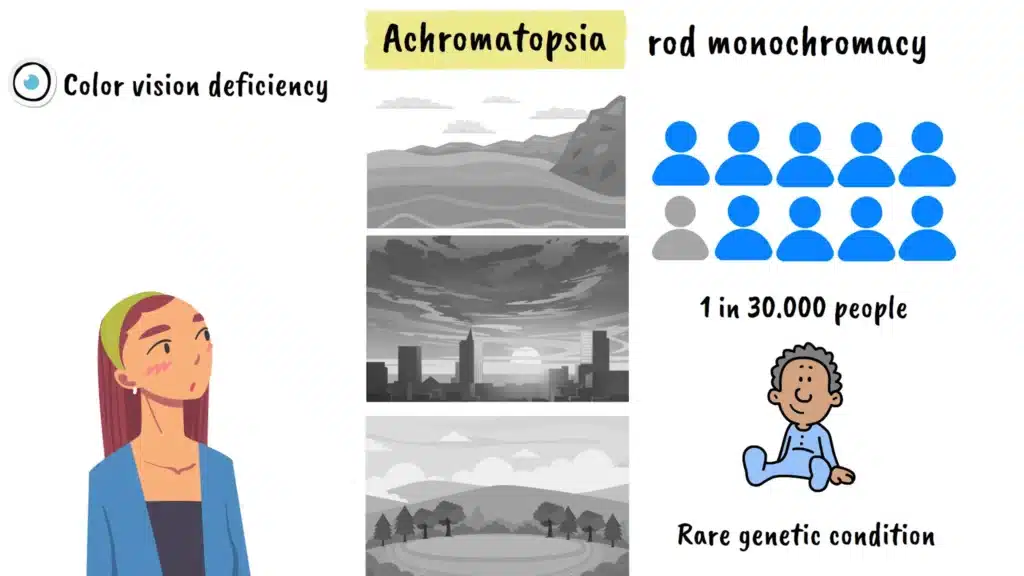
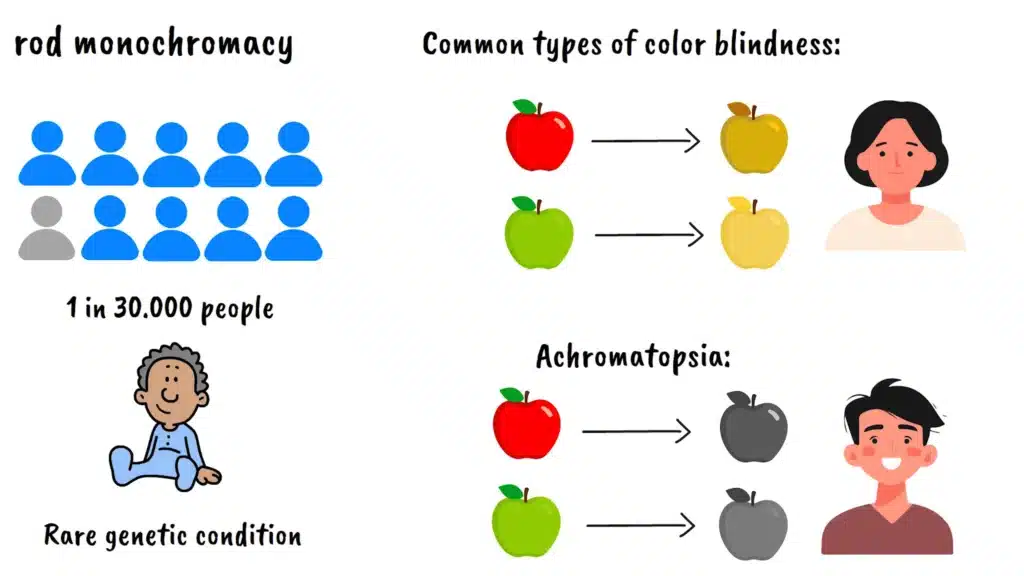
Achromatopsia, also known as rod monochromacy, represents one of the most severe forms of color vision deficiency. Unlike the more common types of color blindness where people have difficulty distinguishing between certain colors like red and green, individuals with achromatopsia experience complete or near-complete absence of color vision. But the challenges extend far beyond simply not seeing colors. This condition affects multiple aspects of vision and daily functioning in ways that many people never consider.
How Vision Works
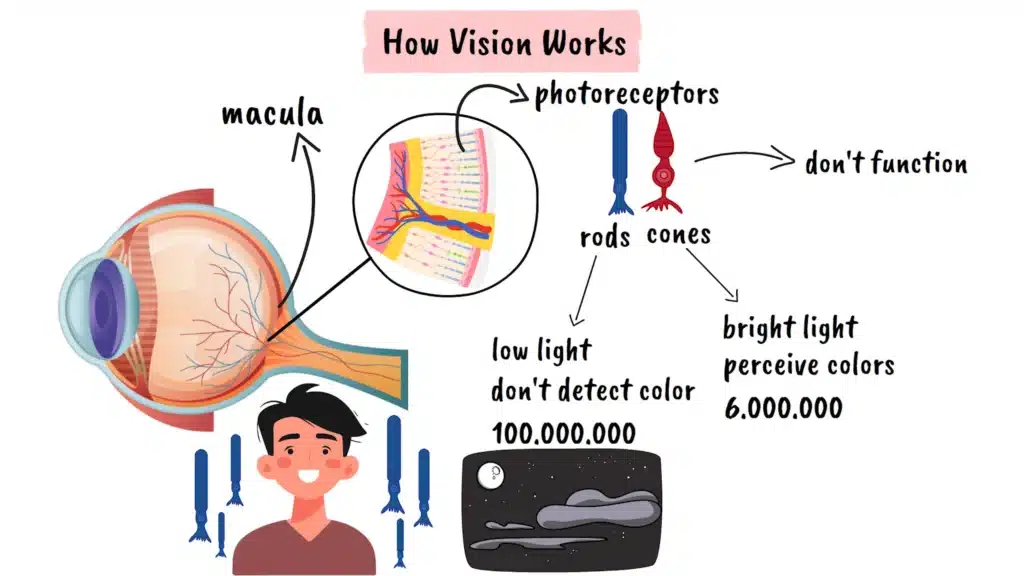
The retina, the light-sensitive tissue at the back of our eyes, contains specialized cells called photoreceptors. There are two main types: rods and cones. Rods are responsible for vision in low light conditions and don’t detect color, while cones enable us to see in bright light and are entirely responsible for our ability to perceive colors. In a typical eye, there are approximately six million cone cells concentrated primarily in the central region called the macula, and about one hundred million rod cells distributed mainly around the periphery.
In achromatopsia, the cone photoreceptors either don’t function at all or function very poorly. This means that people with this condition must rely almost entirely on their rod vision system. Since rods are designed for low-light conditions and become saturated in bright light, this creates a cascade of visual challenges that go well beyond color perception.
Achromatopsia Symptoms
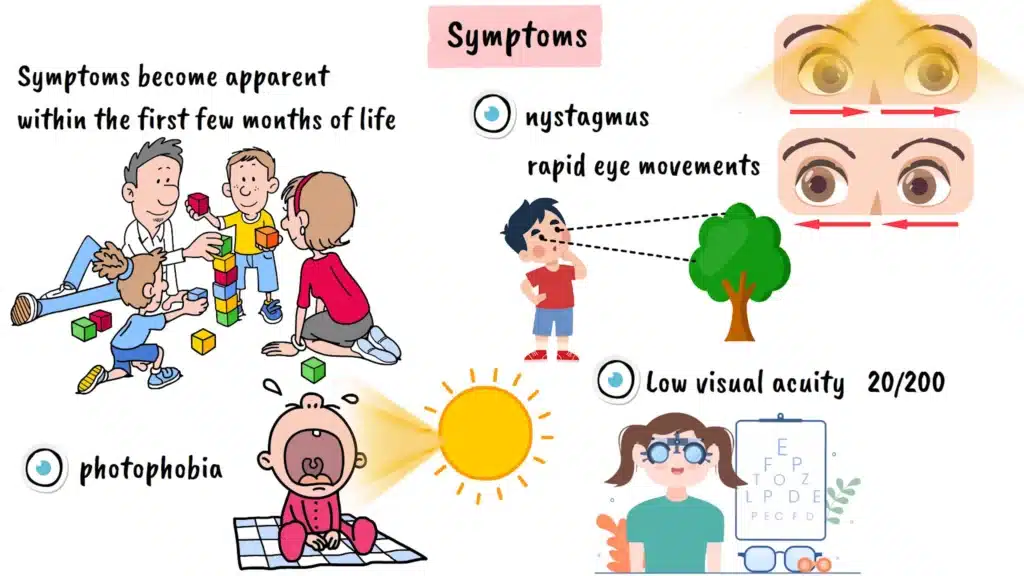
The symptoms of achromatopsia typically become apparent within the first few months of life, though parents might not immediately recognize what they’re observing. One of the earliest and most noticeable signs is photophobia, an extreme sensitivity to light. Babies with achromatopsia often cry or become distressed in bright environments, preferring dimly lit spaces. They may squint excessively or turn away from light sources that wouldn’t bother other infants.
Another early indicator is nystagmus, which refers to involuntary, rapid eye movements that appear as a constant trembling or shaking of the eyes. This occurs because the visual system is trying to compensate for the poor central vision by constantly searching for a clearer image. The nystagmus is typically most pronounced when trying to focus on distant objects and tends to worsen in bright lighting conditions.
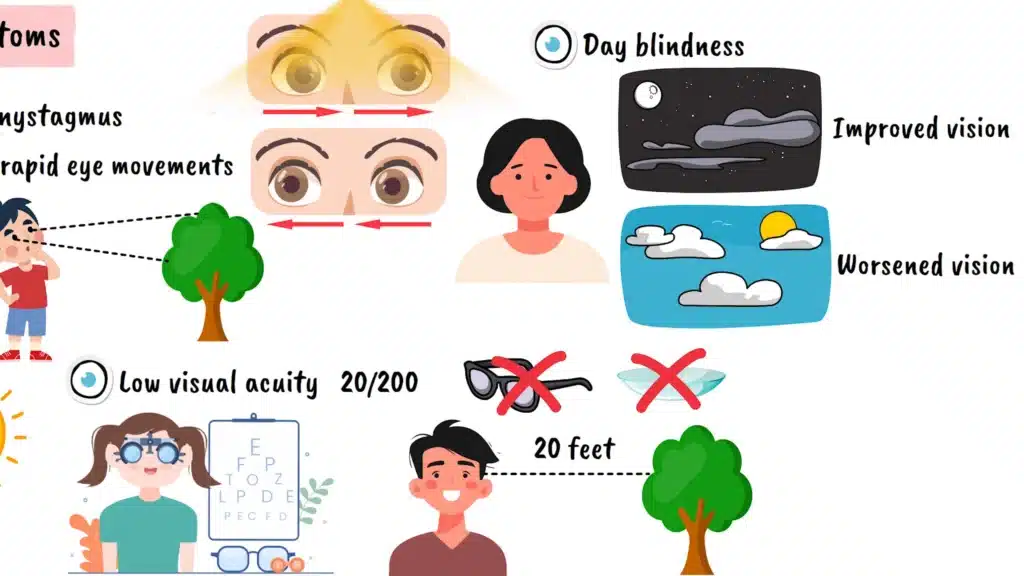
Visual acuity, or the sharpness of vision, is significantly reduced in people with achromatopsia. Most individuals with complete achromatopsia have visual acuity around twenty over two hundred, which means they need to be twenty feet away from something that a person with normal vision can see clearly from two hundred feet away. This level of vision impairment cannot be corrected with conventional glasses or contact lenses because it stems from the fundamental dysfunction of the cone cells rather than refractive errors.
Many people with achromatopsia also experience day blindness, or hemeralopia. Their vision actually improves in dimmer lighting conditions because their rod cells, which are naturally more sensitive to light, can function more effectively without being overwhelmed by brightness. This creates a counterintuitive situation where someone might navigate better at dusk than at midday.
Achromatopsia Causes and Genetics
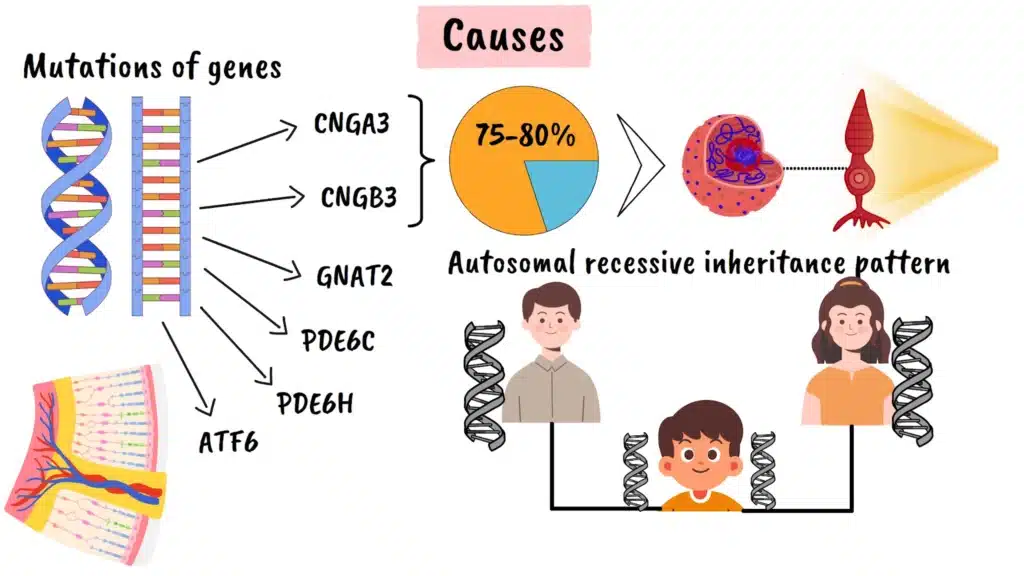
The genetic basis of achromatopsia involves mutations in any of six different genes. The most commonly affected genes are CNGA3 and CNGB3, which together account for approximately seventy-five to eighty percent of all cases. These genes provide instructions for making proteins that are essential components of the cone cell’s ability to respond to light. Additional genes associated with the condition include GNAT2, PDE6C, PDE6H, and ATF6, each playing crucial roles in the cone photoreceptor signaling pathway.
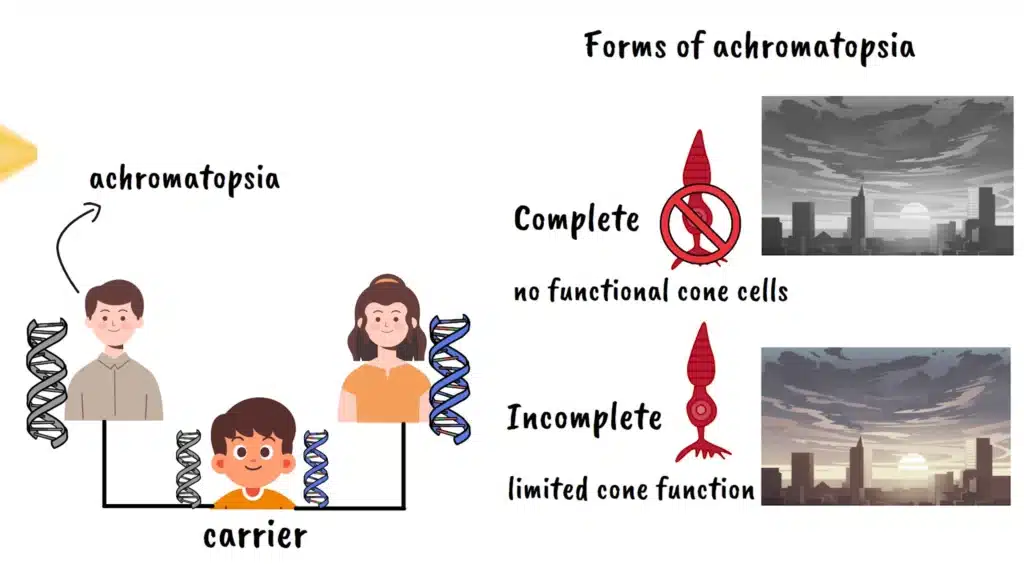
Achromatopsia follows an autosomal recessive inheritance pattern. This means that an individual must inherit a mutated copy of the gene from both parents to develop the condition. Parents who each carry one mutated copy typically show no symptoms themselves but have a twenty-five percent chance with each pregnancy of having a child with achromatopsia. Interestingly, every child of someone with achromatopsia will be a carrier of the condition, though they won’t be affected unless their other parent is also a carrier.
There are two main forms of achromatopsia: complete and incomplete. People with complete achromatopsia have no functional cone cells and see the world entirely in grayscale. Those with incomplete achromatopsia retain some limited cone function and may be able to perceive very faint colors under specific lighting conditions, though their color vision is still severely impaired.
Achromatopsia Diagnosis
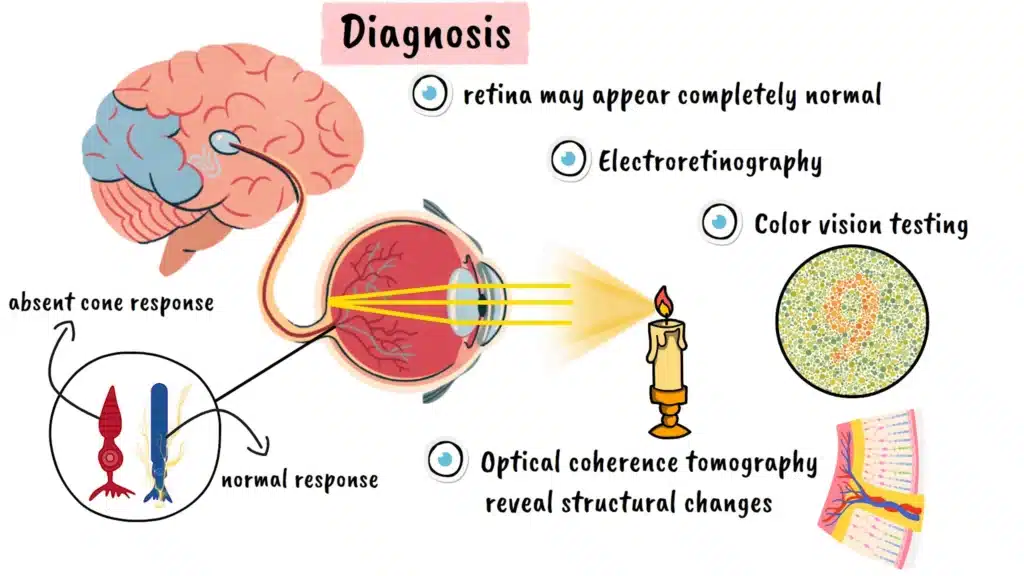
Diagnosing achromatopsia requires a comprehensive approach that goes beyond standard eye examinations. While the retina may appear completely normal during a routine fundus examination, specialized testing reveals the underlying cone dysfunction. Electroretinography, which measures the electrical responses of retinal cells to light stimulation, shows absent or severely reduced cone responses while rod responses remain normal. Color vision testing demonstrates the extent of color discrimination loss, and optical coherence tomography can reveal structural changes in the retinal layers where cone cells are concentrated.
Achromatopsia Treatment Options
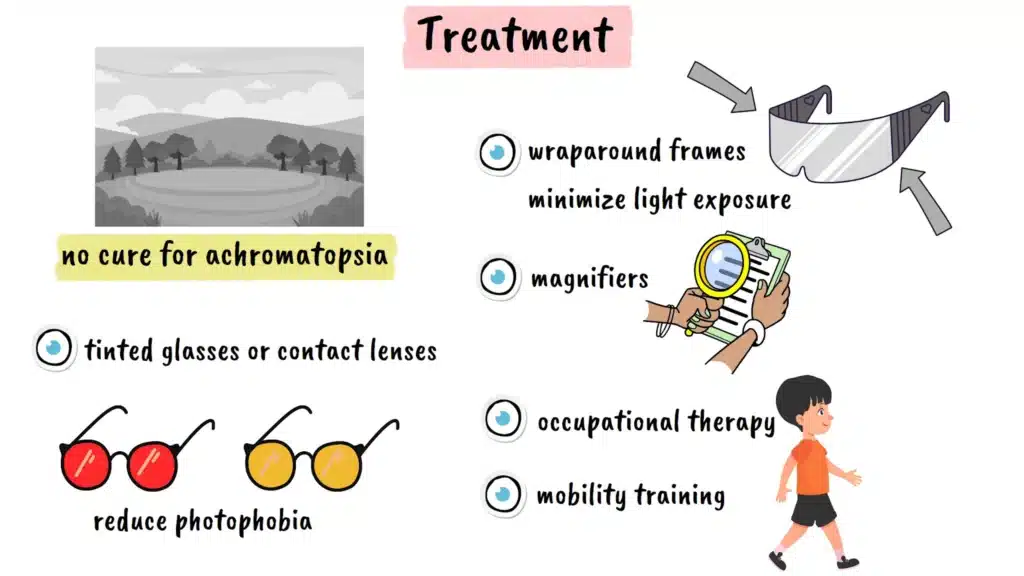
Currently, there is no cure for achromatopsia, but several management strategies can significantly improve quality of life. The most common intervention involves specially tinted glasses or contact lenses, particularly those with red or amber filters. These lenses help reduce photophobia by filtering out the wavelengths of light that are most uncomfortable for people with the condition. Some individuals benefit from wraparound frames with additional side shields to minimize light exposure from all angles.
Low vision aids such as magnifiers can help compensate for reduced visual acuity, particularly for close-up tasks like reading. Many people with achromatopsia also benefit from occupational therapy to learn adaptive techniques for daily activities and mobility training to navigate safely in various environments.
Achromatopsia Daily Life Challenges
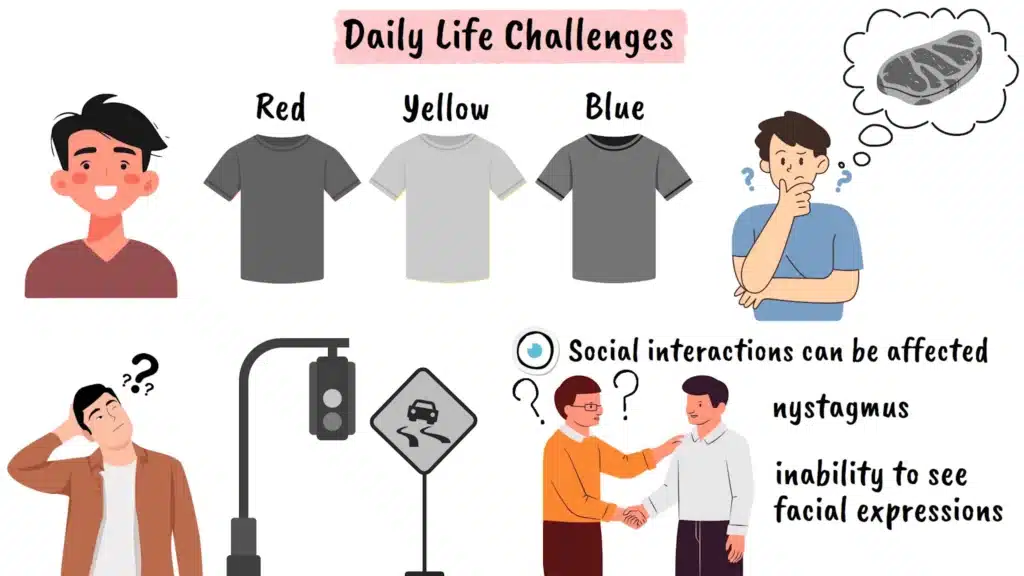
The daily challenges faced by people with achromatopsia extend far beyond the medical symptoms. Simple activities that most people take for granted become complex problems requiring creative solutions. Choosing appropriate clothing becomes difficult without color cues, leading many to organize their wardrobes by location or ask family members to help coordinate outfits. Cooking presents multiple challenges, from determining if meat is properly cooked to distinguishing between similar-looking ingredients.
Technology use can be particularly challenging, as many interfaces rely heavily on color coding to convey information. Traffic lights, warning signs, and electronic displays often become difficult to interpret. Many individuals develop personal strategies, such as memorizing the positions of traffic light colors or relying on other contextual cues.
Social interactions can also be affected by the condition. The involuntary eye movements associated with nystagmus may make some people self-conscious, and the inability to see facial expressions clearly in bright lighting can impact communication. Many individuals report that others don’t fully understand the extent of their visual challenges, leading to feelings of isolation or misunderstanding.
Achromatopsia Research and Gene Therapy
Despite these challenges, research into potential treatments for achromatopsia has advanced dramatically in recent years. Gene therapy has emerged as the most promising approach, with multiple clinical trials currently underway or recently completed. The concept is relatively straightforward: use a harmless virus to deliver a normal copy of the defective gene directly to the cone cells in the retina.
Several phase one and phase two clinical trials have been conducted, with mixed but encouraging results. Some participants have shown improvements in light sensitivity, visual acuity, and even limited color perception. Particularly promising are results from trials involving children, where the brain’s natural plasticity during development appears to enhance the therapy’s effectiveness. Recent studies have demonstrated that gene therapy can actually reactivate dormant cone signaling pathways in the brain, creating visual experiences that these children had never had before.
The timing of treatment appears to be crucial. Research suggests that younger patients, particularly those treated during childhood or adolescence, may experience better outcomes than adults. This is likely due to the brain’s greater ability to adapt and form new neural connections during development. However, gene therapy trials are ongoing, and researchers continue to refine techniques and identify optimal treatment windows.
Achromatopsia Future Outlook
Beyond gene therapy, other innovative approaches are being investigated. These include the use of specially designed contact lenses that can enhance contrast, digital aids that provide audio feedback about colors, and pharmaceutical interventions that might support cone cell function.
The progress in achromatopsia research reflects broader advances in understanding inherited retinal diseases. As genetic testing becomes more sophisticated and accessible, earlier and more precise diagnosis becomes possible. This, combined with advancing gene therapy techniques, offers genuine hope for the future.
For families affected by achromatopsia, genetic counseling provides valuable information about inheritance risks and family planning options. Support organizations and online communities offer practical advice, emotional support, and advocacy for research funding and accessibility improvements.
Conclusion
Living with achromatopsia requires adaptability, creativity, and often a strong support network. While the condition presents significant challenges, many individuals lead fulfilling, independent lives by developing effective coping strategies and leveraging available assistive technologies. The key lies in understanding the condition thoroughly, implementing appropriate management strategies, and staying informed about emerging treatments.
As our understanding of achromatopsia continues to evolve and treatment options expand, there is increasing reason for optimism. The combination of advancing gene therapy techniques, improved diagnostic methods, and greater awareness of the condition’s impact on daily life is creating new possibilities for those affected by this challenging but treatable genetic condition. The future holds promise for not just managing achromatopsia, but potentially restoring the full spectrum of human color vision.